Simple Synthetic Strategy Converts Blue-Emissive Molecules into Multicolor Luminescent Materials
- Research
Researchers demonstrate stimuli-responsive luminescence in dumbbell-shaped Zn(II) complexes with extended terminals
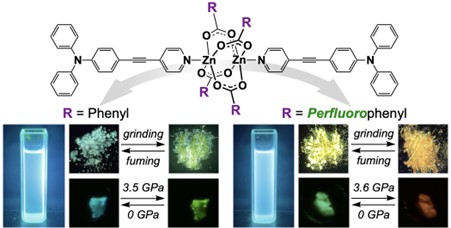
Researchers have demonstrated that bridging π-conjugated emissive chromophores with an aromatic fluorinated Zn(II) paddlewheel framework facilitates wide-ranging and reversible luminescence color modulation in the solid state in the presence of mechanical stress and hydrostatic pressure. This step converts a blue-emissive small organic molecule into a multicolor luminescent material. In solution, it behaves as a blue emitter, whereas in the solid state, its emission color changes reversibly over a wide range in response to the environment.
Chemistry involves the fundamental interplay between the structures and properties of molecules. Notably, subtle changes in molecular structure and crystal packing can be amplified into macroscopic phenomena such as optical responses.
Zn(II) is an earth-abundant and low-toxicity metal, and paddlewheel-type Zn(II) dimers are well-established structural motifs. They are traditionally regarded as electronically silent structural units. Recently, a study led by Professor Akiko Hori from the Shibaura Institute of Technology, Japan, including Mr. Yuta Takeuchi from the same institute, and Prof. Yoshiki Ozawa and Prof. Masaaki Abe from the University of Hyogo, Japan, hypothesized that combining this flexible metal-carboxylate scaffold with π-extended emissive ligands and aromatic fluorination could unlock new, adaptive excited-state behavior under external stimuli. Advancing this work, they observed a striking contrast between the fluorinated and non-fluorinated crystals—particularly the much larger and smoother color shifts observed under mechanical grinding and hydrostatic pressure. This motivated them to conduct a deeper exploration of how structural flexibility, intermolecular interactions, and external forces cooperate to control luminescence.
This curiosity ultimately led to the discovery of wide-range, reversible color modulation in a simple molecular crystal system. The novel findings of the research group were published in the Inorganic Chemistry Frontiers journal as a Front Cover article on January 19, 2026.
Prof. Hori highlights the central message of their study: “A key point of this study is that a small blue-emissive molecule can be converted into a multicolor luminescent material in a one-step reaction. The resulting material shows reversible color changes over a wide range, from blue in solution to green and red in the solid state, depending on the environment.”
By introducing pentafluorobenzoate bridges into a dumbbell-shaped Zn(II) dimer, the researchers created a molecular crystal that exhibits continuous and nearly reversible emission color changes, spanning from green to orange-red upon gentle grinding or compression. Notably, these changes occur without obvious chemical degradation and are instead attributed to pressure-induced structural flexibility and adaptive intermolecular interactions within a simple Zn₂(μ-carboxylate)₄ framework.
The reversible and visually distinct luminescence changes observed in this study demonstrate how mechanical stress and pressure can be directly translated into optical responses in molecular crystals. In particular, the continuous color modulation in the solid state provides insight into how structural flexibility and intermolecular interactions govern stimulus-responsive luminescence. Because the emission color changes in response to mechanical force, pressure, and the surrounding molecular environment, the material may help visualize changes that are otherwise difficult to see. In the future, this concept may lead to luminescent materials that indicate pressure, strain, or molecular interactions through color changes.
“This research addresses a fundamental challenge in materials chemistry: how to achieve broad, reversible, and controllable color modulation in solid-state luminescent materials using simple and chemically robust molecular components. By demonstrating that aromatic fluorination markedly enhances structural flexibility and pressure adaptability, this study reveals how subtle modifications to a Zn(II)-based framework can strongly influence excited-state behavior in molecular crystals. These findings deepen our understanding of stimulus-responsive luminescence and structure–property relationships, and may help guide the design of adaptive optical materials,” says Mr. Takeuchi.
Overall, this study presents a simple design concept for converting blue-emissive molecules into multicolor luminescent materials and may contribute to the development of new optical materials that communicate environmental and structural changes through light.
Reference
|
Title of original paper: |
Multicolor and reversible stimuli-responsive luminescence of dumbbell-shaped Zn(II) complexes with extended triphenylamine-attached ethynylpyridine terminals |
|
Journal: |
Inorganic Chemistry Frontiers |
|
DOI: |
Additional information for EurekAlert
| Latest Article Publication Date: | 19 January 2026 |
| Method of Research: | Experimental study |
| Subject of Research: | Not applicable |
| Conflicts of Interest Statement: | There are no conflicts to declare. |
Authors
About Shibaura Institute of Technology (SIT), Japan
Shibaura Institute of Technology (SIT) is a private university with campuses in Tokyo and Saitama. Since the establishment of its predecessor, Tokyo Higher School of Industry and Commerce, in 1927, it has maintained “learning through practice” as its philosophy in the education of engineers. SIT was the only private science and engineering university selected for the Top Global University Project sponsored by the Ministry of Education, Culture, Sports, Science and Technology and had received support from the ministry for 10 years starting from the 2014 academic year. Its motto, “Nurturing engineers who learn from society and contribute to society,” reflects its mission of fostering scientists and engineers who can contribute to the sustainable growth of the world by exposing their over 9,500 students to culturally diverse environments, where they learn to cope, collaborate, and relate with fellow students from around the world.
Website: https://www.shibaura-it.ac.jp/en/
About Professor Akiko Hori from SIT, Japan
Dr. Akiko Hori is a Professor at the Graduate School of Engineering and Science and the Undergraduate School of Engineering, both at Shibaura Institute of Technology, Japan. She leads the Laboratory of Molecular Assemblies, which aims to manipulate intermolecular forces to create highly tunable crystal space groups. Specifically, her group explores the creation of dynamic crystal fields using fluorine-substituted metal complexes, the synthesis and functional characterization of molecular nanowire variants with different metals, and the synthesis and investigation of the luminescence behavior of bipyridine derivatives.
Funding information
This work was supported by S-SPIRE project of Shibaura Institute of Technology (Akiko Hori). Masaaki Abe acknowledges financial support from Special Research Projects Grant, University of Hyogo (FY2025). Yoshiki Ozawa acknowledges financial support for Grant-in-Aids for Scientific Research C, 22K05147, of JSPS KAKENHI.
Image
Title: Dumbbell-shaped Zn(II) complexes with extended triphenylamine-attached ethynylpyridine terminals
Caption: Researchers explore the multicolor and reversible stimuli-responsive luminescence behavior of these exciting molecules.
Credit: Professor Akiko Hori from Shibaura Institute of Technology, Japan
Source link: https://pubs.rsc.org/en/Content/ArticleLanding/2026/QI/D5QI02451J
License type: CC BY 3.0
Usage restrictions: Credit must be given to the creator.
Media contact: Kohei Tsuchiya
E-mail: koho@ow.shibaura-it.ac.jp