Scientists Synthesize Isotopic Atropisomers Based on Carbon Isotope Discrimination
- Research
The novel quinazolin-4-one derivatives with the isotopic atropisomerism exhibited high rotational stability and stereochemical purity
Reaction mechanisms in synthetic organic chemistry and medicinal chemistry require fundamental research on isotopically chiral compounds. Although the synthesis of isotopic atropisomers based on hydrogen/deuterium discrimination has been previously reported, isotopic atropisomerism based on the discrimination of carbon isotopes remains undetected. Recently, researchers have demonstrated the asymmetric synthesis of quinazolin-4-one derivatives with carbon isotopic atropisomerism, which exhibited high rotational stability and stereochemical purity.
Synthesis and characterization of novel isotopic atropisomers
Researchers demonstrate the asymmetric synthesis of quinazolin-4-one derivatives possessing isotopic atropisomerism based on carbon isotope discrimination. The synthesized molecules exhibited high rotational stability and stereochemical purity.
Credit:Professor Osamu Kitagawa from Shibaura Institute of Technology, Japan
In chemistry, a molecule or ion is said to be chiral if it cannot be superposed on to its mirror image by any combination of rotations, translations, or conformational changes. A chiral molecule or ion exists in two forms, called enantiomers, that are mirror images of each other; they are often distinguished as either ‘right-handed’ or ‘left-handed’ by their absolute configuration. Enantiomers exhibit similar physical and chemical properties, except when interacting with polarized light and reacting with other chiral compounds, respectively.
In recent years, isotopically chiral compounds, based on the discrimination of isotopes of an element, have generated considerable interest in structural and synthetic organic chemistry, medicinal chemistry, and fundamental reaction mechanisms due to their novel properties and potential applications. Numerous optically active isotopic molecules with an asymmetric carbon have been synthesized based on hydrogen/deuterium (H/D) discrimination. However, the synthesis, detection, and characterization of isotopic atropisomers—stereoisomers resulting from hindered rotation about single bonds, wherein the steric strain barrier to rotation is high enough to allow for the isolation of the conformers—is particularly challenging. To date, a handful of such molecules based on H/D discrimination have been reported.
Recently, a team of researchers, led by Professor Osamu Kitagawa of the Department of Applied Chemistry at the Faculty of Engineering at the Shibaura Institute of Technology in Japan, has successfully demonstrated the asymmetric synthesis of isotopic atropisomers based on the discrimination of carbon isotopes. Their work was made available online in The Journal of Organic Chemistry on 10 June 2023. It was co-authored by Ryunosuke Senda and Yuka Watanabe, graduate students at the Department of Applied Chemistry, Shibaura Institute of Technology.
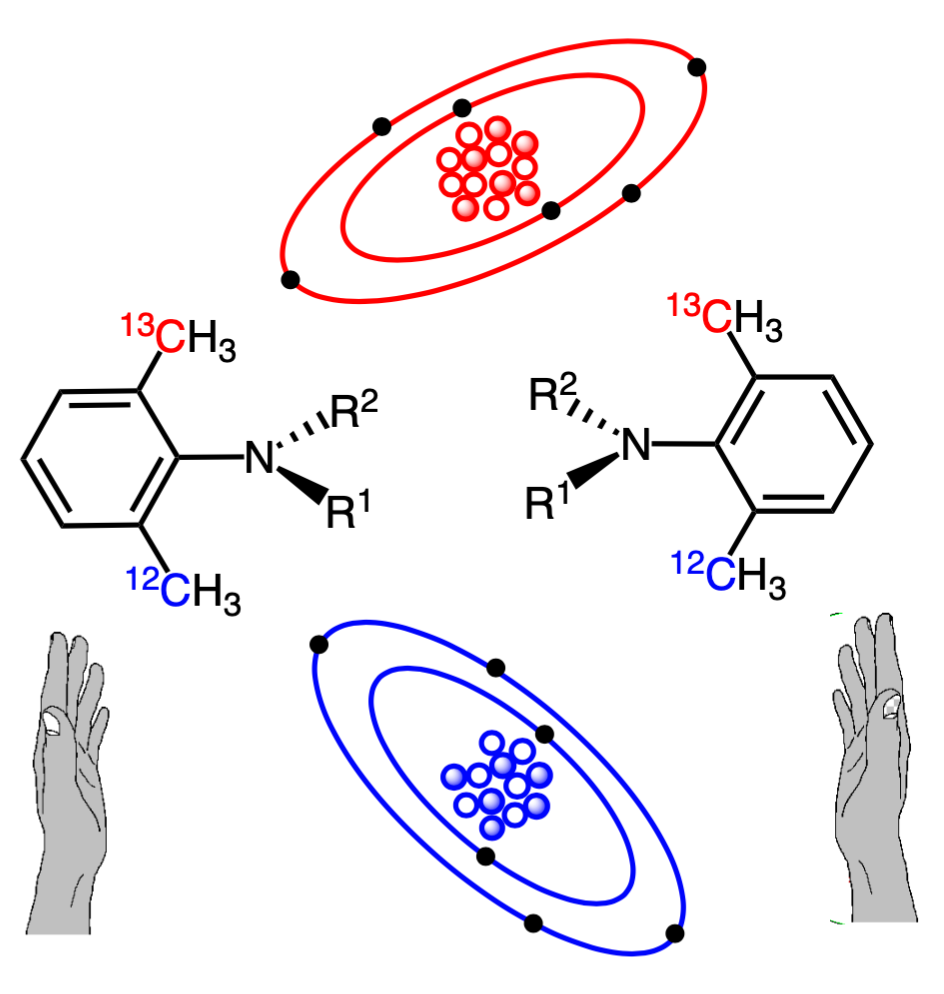
Taking inspiration from their previous work (J. Org. Chem. 2022, 87, 13501) on the synthesis of CH3/CD3-atropisomeric quinazolin-4-one derivative, the team successfully prepared both enantiomers of 2-ethyl quinazolin-4-one with isotopic atropisomerism (N–C axial chirality) based on ortho-12CH3/13CH3 discrimination. They achieved this feat via an asymmetric synthesis method involving Suzuki–Miyaura cross-coupling. “The synthesized isotopic atropisomers based on 12C/13C discrimination were cryptochiral compounds having no optical rotation,” points out Prof. Kitagawa.
Since the researchers could not verify the existence of isotopic atropisomerism in the above-mentioned molecules, they further synthesized diastereomeric 3-aryl quinazolin-4-one derivatives possessing an asymmetric carbon atom along with isotopic atropisomerism. The diastereomers could be clearly distinguished (and the isotopic atropisomerism could be verified) using 1H and 13C nuclear magnetic resonance. They also exhibited high enantiomeric and diastereomeric stereochemical purity and rotational stability. Therefore, diastereomeric 3-aryl quinazolin-4-ones are an excellent scaffold for verifying diverse isotopic atropisomers.
Taken together, the novel findings of this research are bound to enrich our fundamental understanding of isotopic atropisomers, with positive implications in organicand medicinal chemistry. “This work reports the first isotopic atropisomers based on 12C/13C discrimination, which could potentially kindle academic curiosity in researchers to conduct similar studies in fundamental organic chemistry,” notes an optimistic Prof. Kitagawa.
Reference
| Title of original paper: | Synthesis of Isotopic Atropisomers Based on 12C/13C Discrimination |
| Journal | The Journal of Organic Chemistry |
| Article link: | 10.1021/acs.joc.3c01004 |
Funding Information
This work was partly supported by a research grant from Nagase Science and Technology Foundation.